Author: Sam Antrobus | Posted On: 24 Nov 2025
Two breakthrough dementia treatment options approved overseas cannot get Pharmaceutical Benefits Scheme (PBS) funding in Australia, leaving patients without access. Donanemab and Lecanemab slow early Alzheimer’s disease by roughly 30% and are already available in the US and EU. But despite approval from the Therapeutic Goods Administration (TGA), they remain unfunded on the PBS and inaccessible to Australian patients. This happens because the evidence produced in dementia trials does not line up with what the PBS needs to see to justify funding, meaning that promising treatments can clear regulatory approval yet remain out of reach for Australians.
PBS Standards Require Evidence That Dementia Trials Struggle to Provide
These drugs work by clearing amyloid plaques from the brain, a biological process confirmed through PET scans. However, the PBS does not reimburse medicines for biological changes alone, it funds them when they produce clear, measurable improvements in daily functioning, independence and quality of life. This is where dementia treatments fall short: modest benefits, slow progression and difficulties translating short-term improvements on cognitive tests into long-term outcomes.
Challenge 1: Quality-of-Life Benefits Are Undercaptured
Traditional quality-of-life questionnaires such as the EQ-5D were not built for dementia and fail to capture key disease-specific impacts such as behavioural changes, loss of insight and declining independence. When the measurement tools fail to capture the real impact of the disease, the PBS model underestimates the benefits and the treatments look less effective than they actually are.

Challenge 2: Caregiver Impact Is Missing From the Evidence
Dementia affects two people at once: the patient and the caregiver. However, carer health impacts, work disruption and emotional burden are often excluded from PBS assessments. Excluding caregiver outcomes removes a major source of economic value, making the drugs appear to deliver fewer benefits than they would in the real world, another hit to PBS cost-effectiveness scores.
Challenge 3: Infrastructure Costs Shift the Economics
These treatments rely on costly infrastructure: PET scans, genetic tests, regular MRIs and infusion clinics. Pharmaceutical Benefits Advisory Committee (PBAC) assessments depend heavily on long-term predictions about whether the drugs meaningfully delay aged-care entry and reduce overall system costs. Small uncertainties in those long-term predictions can swing the model, quickly turning a treatment from cost-effective to not, and making PBS approval harder even when clinical benefits are real.
Australia’s Recent Decisions
This is now reflected in real regulatory outcomes:
- Donanemab: TGA approved with strict monitoring requirements (mandatory MRIs and confirmed amyloid pathology), but the PBAC declined PBS listing in July 2025 due to risk-benefit uncertainty.
- Lecanemab: Initially rejected by TGA in October 2024 and later approved in September 2025 with added safety data; PBS funding is still pending, with mandatory MRI surveillance and recommended genetic screening.
International Comparisons
Health systems globally are reaching similar conclusions, even when regulators approve the drugs.
- US: Medicare covers both, though use of Lecanemab comes with mandatory participation in a real-world evidence registry.
- EU: Lecanemab is approved and donanemab has received a positive opinion from regulators, though neither currently receive widespread funding.
- UK: Although approved by the Medicines and Healthcare products Regulatory Agency (MHRA), the National Institute for Health and Care Excellence (NICE) has declined both drugs for NHS funding due to cost-effectiveness concerns. The core reasons for the NICE decision were that the benefits (slowing disease progression by a small amount) did not justify the significant cost of the drug, the frequent hospital infusions, and the intensive monitoring required for side effects.
The following timelines illustrate how Australia’s regulatory pathway compares to other major markets
Chart 1: Donanemab approval timeline following Phase III trial results across the US, UK, EU and Australia
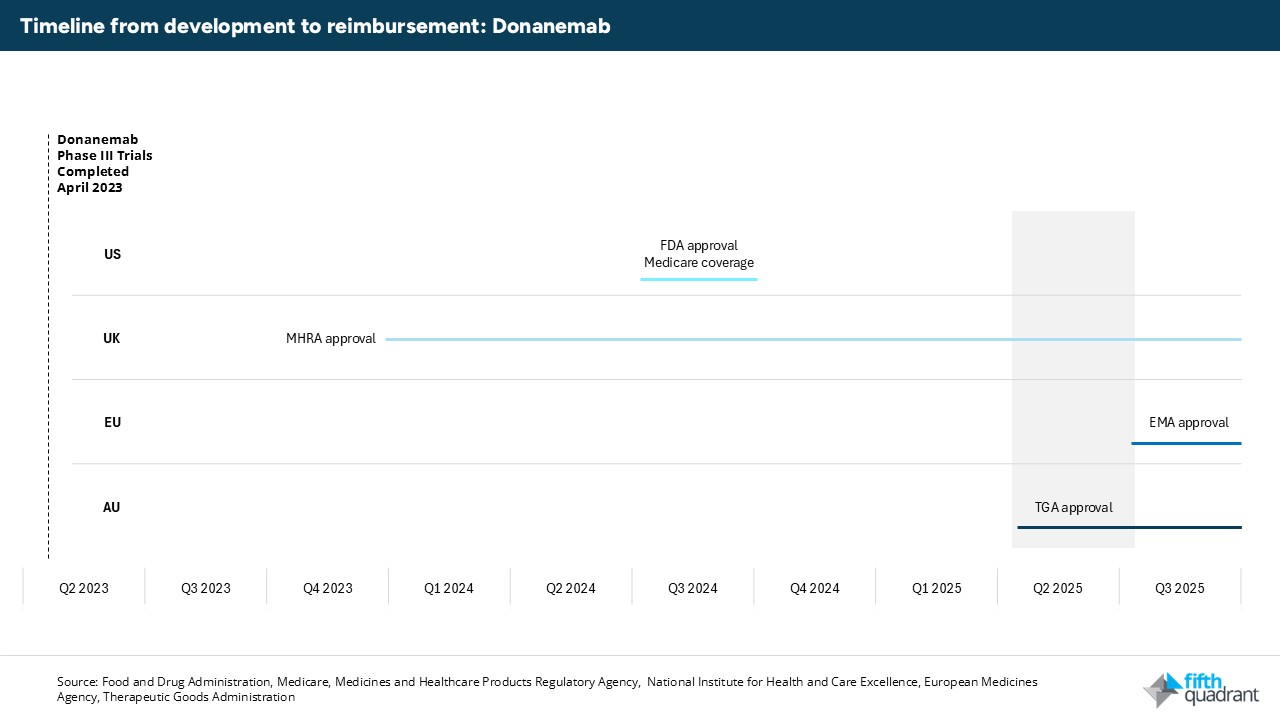
Chart 2: Lecanemab approval timeline following Phase III trial results across the US, UK, EU and Australia
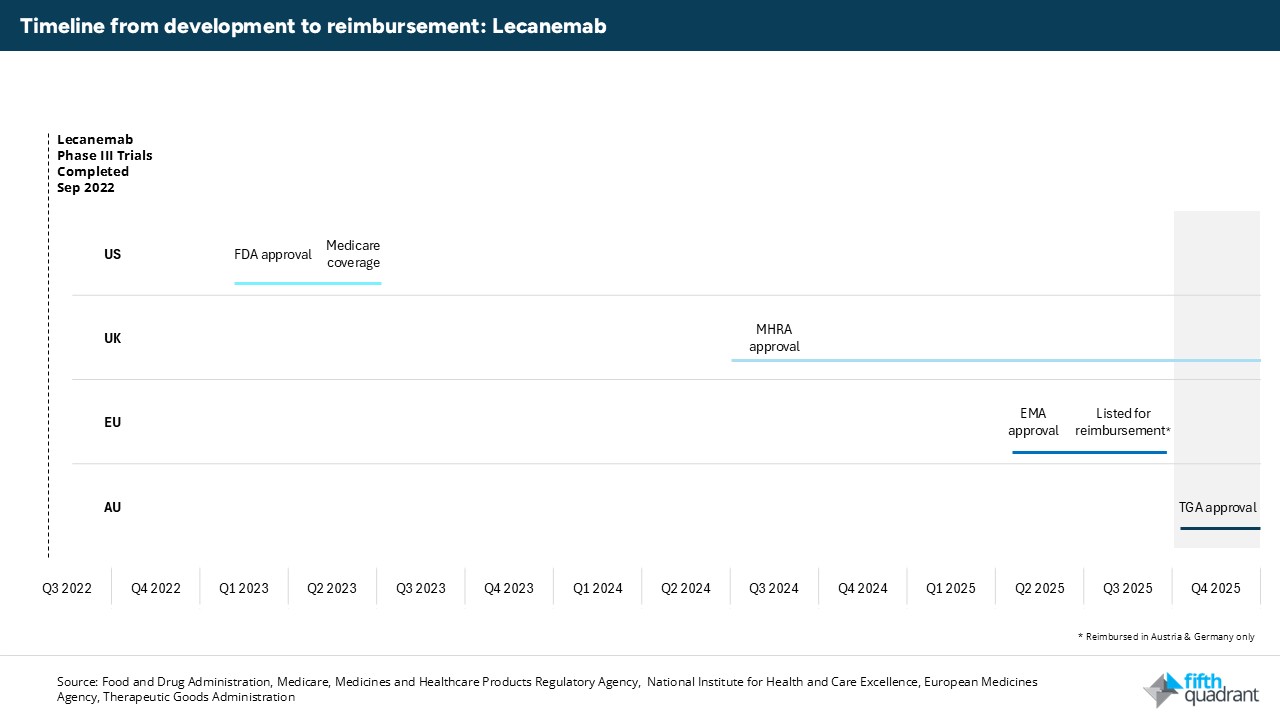
These timelines reinforce that, whilst Australia is slower to react in approving new dementia treatments for use, regulatory approval does not guarantee reimbursement and the challenge for donanemab and lecanemab resonates internationally.
What Pharmaceutical Companies Need to Demonstrate for PBS Approval
To progress these treatments through PBAC, sponsors must rethink their evidence strategy:
1. Build Australian-specific long-term outcome models
These models need to link early cognitive changes to meaningful outcomes such as delayed aged-care entry, extended independence and reduced caregiver hours.
2. Elevate caregiver impacts to core evidence
Submissions should quantify reduced caregiver health costs, reduced lost work hours and the economic value of delaying full-time care.
3. Pursue risk-sharing pricing agreements
PBAC is more receptive when companies share uncertainty through pricing models that limit financial risk to the health system.
- Real-world evidence commitments: Agreeing to collect Australian patient data after launch helps demonstrate the treatment delivers its expected benefits.
- Milestone-based pricing: The government only pays full price if the drug achieves agreed clinical or functional outcomes.
- Safety-linked cost caps: Pricing is capped if safety issues or early discontinuation reduce the expected duration of treatment.
- Budget-impact controls: Sponsors can set volume or expenditure limits that prevent total system costs from exceeding agreed thresholds.
- Temporary introductory pricing: Offering lower initial pricing allows PBS to fund earlier while more evidence is generated.
4. Provide system-readiness implementation plans
Sponsors must demonstrate how PET scan access, MRI capacity, genetic testing and infusion services will be managed to avoid bottlenecks and cost overruns.
Looking Forward
The science behind Alzheimer’s disease treatments is progressing rapidly, but access will depend on evidence that reflects how dementia affects patients, families and the broader system, not just biomarker changes.
The question now is whether pharmaceutical companies will adapt their evidence strategies to Australia’s requirements, or whether Australian patients will continue waiting for access.
At Fifth Quadrant, we help organisations make sense of the forces shaping Australia’s health system. Our research brings clarity to emerging treatments, policy decisions and their real-world impact. To learn more about our work in the healthcare sector, click here or contact us today.
Posted in Healthcare

