Author: Sam Antrobus | Posted On: 06 Mar 2026
Some of the most important medicines in use today began their lives treating sheep, cattle, or companion animals. That is not a coincidence. The relationship between animal health and human medicine, referred to by the WHO as ‘One Health’, is deep and structural, and a growing body of evidence suggests it is being more deliberately put to work than ever before.
The basic idea is straightforward: humans and animals share a great deal of biology. That means treatments developed for animals can, under the right conditions, become the starting point for human medicines. The question researchers and drug developers are increasingly asking is: when, and how reliably, does that happen?
A proven pattern, not a lucky accident
The clearest examples come from treatments for parasitic diseases, illnesses caused by worms and other organisms that affect hundreds of millions of people in tropical and developing regions. Ivermectin, originally developed to protect livestock from parasites, was later approved to treat a blinding disease in humans called river blindness. Animal use began in 1981; human approval in the US followed in 1996. Moxidectin, another livestock treatment, spent more than 20 years in veterinary use before the FDA approved human use in 2018.
What made both transitions possible was the fact that the biology of the target, the parasite, is similar whether it is living in an animal or a person. The years of veterinary use also generated a large body of real-world safety information, which helped researchers design human trials with greater confidence.
Albendazole, a deworming drug familiar to pet owners, followed a similar path. It was developed inside an animal health company, tested in worm-infected sheep, and only later adapted for use in human deworming programmes. As the WHO notes, the case for human development was partly built on evidence that had accumulated through agricultural and veterinary use first.

Timeline showing key veterinary-origin drugs (ivermectin, moxidectin, albendazole) and the approximate gap in years between animal approval and human approval.
What is coming next
Several animal treatments are now being tested for human use. Emodepside, a dewormer for cats and dogs, is currently in human trials for worm infections, with early results published in leading medical journals. A veterinary drug called oxfendazole has completed its first human safety trials, targeting a serious brain infection caused by tapeworm larvae. Modern flea and tick treatments are being evaluated for human scabies, a skin condition caused by tiny mites. In each case, years of safe animal use have given researchers a meaningful head start.
Why pets may hold the key to human cancer treatment
Not all of this is about taking a drug from one species and giving it to another. Comparative oncology, the study of cancer across species, takes a different approach. Researchers have found that dogs and cats develop cancers that closely resemble human cancers in how they grow and respond to treatment. That makes them a uniquely useful group for testing new cancer therapies in a realistic clinical setting, rather than in laboratory mice.
The US National Cancer Institute’s Comparative Oncology Program has explicitly called this an underused resource. A 2025 commentary in Nature Reviews Cancer reached a similar conclusion, noting that trials in pet dogs have already helped move promising cancer treatments into early human studies. University veterinary programmes are now running cancer treatment trials in dogs that run alongside equivalent human trials, a model that benefits both the animals being treated and the humans who may eventually benefit from the findings.
The economics behind the science
Developing a new animal drug is significantly cheaper and faster than developing a human one. The Animal Health Institute puts average costs at around US$22.5 million over 6.5 years for a companion-animal drug, compared to estimates for new human drug development that range from US$161 million to US$4.54 billion. That gap creates a practical incentive to establish proof-of-concept in the animal health market first, before committing to the far more expensive human development process.
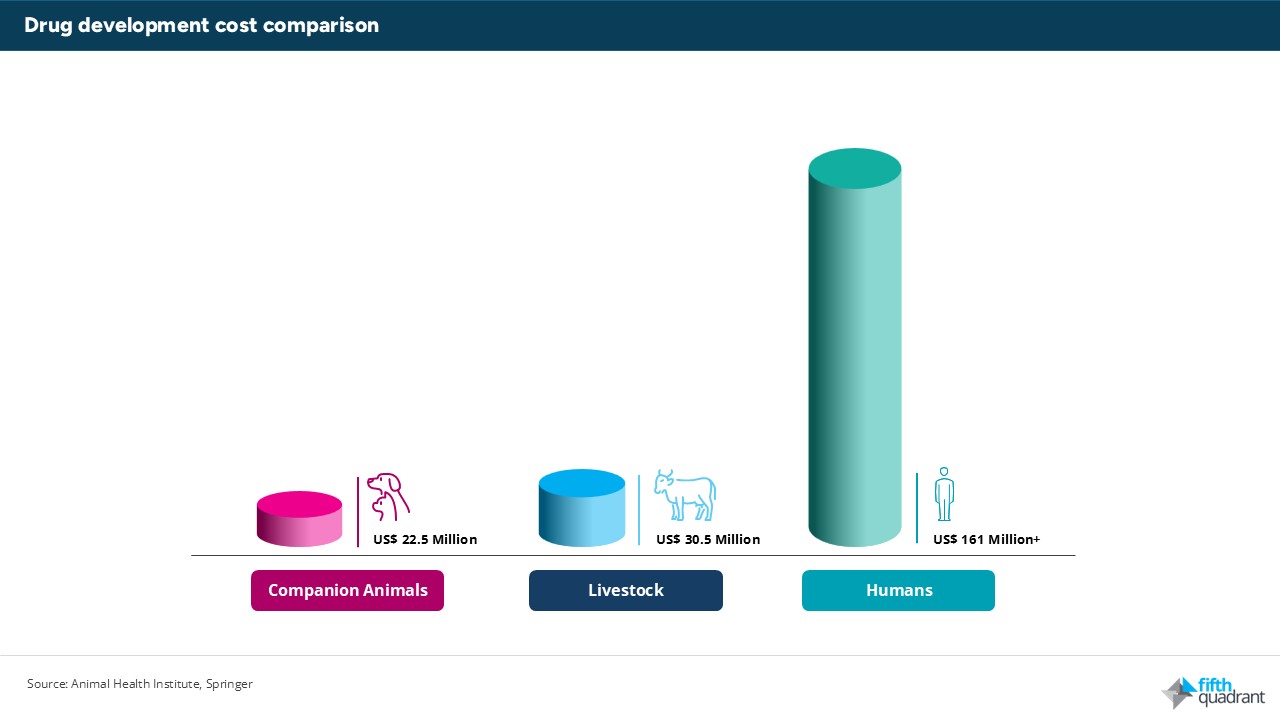
Illustrative comparison of average veterinary vs human drug development cost and timeline
An important line not to cross
Veterinary-to-human translation is a formal, regulated process. An animal medicine does not automatically become a human medicine; separate approval from health regulators is required no matter how long the drug has been used in animals. The warnings issued by the FDA during COVID-19, when people were self-medicating with livestock ivermectin products, are a sharp illustration of why this distinction matters. Veterinary formulations are not tested for use in humans, and the doses can be very different.
Looking forward
The FDA has recently signalled plans to reduce mandatory animal testing for certain types of drugs, looking to replace some of that testing with computer simulations, lab-grown human tissue models, and other newer approaches. The FDA Modernization Act 2.0 is part of the same shift. Over time, this will likely change the shape of the relationship between animal and human medicine: less about animals as a required testing step, and more about animals with naturally occurring disease as one useful source of evidence among several.
The relationship between animal and human medicine is not going away. It is becoming more deliberate.
At Fifth Quadrant, we work across both the animal health and healthcare sectors, helping organisations understand market dynamics, stakeholder needs, and the research questions that matter most. Explore our Animal Health and Healthcare insights, or get in touch to discuss your next project.
Posted in Animal Health, Healthcare

